Abstract
Background: MGTA-117 is a novel anti-CD117 (c-KIT)-amanitin antibody-drug conjugate (ADC) targeting CD117 that is in development as a single agent for myeloid conditioning prior to hematopoietic stem cell transplantation (HSCT). Preclinical studies show rapid and selective depletion of CD117+ stem cells and progenitor cells in bone marrow at low doses in humanized mice and nonhuman primates, rapid and predictable clearance, and potent antileukemic effects in patient-derived xenograft murine models (Lanieri et al, ASH 2020, #1044).
Objectives: To report initial results from Cohort 1 of a phase 1/2 clinical study in which MGTA-117 is administered IV as a single dose in adults with relapsed/refractory (R/R) AML or MDS-EB.
Methods: This is a multicenter, open-label study (NCT05223699). Adult participants (pts) are eligible who have a WHO-defined diagnosis of CD117+ R/R AML or MDS-EB with ≥5% bone marrow blasts (or ≥2% peripheral blasts for MDS-EB); an identified human stem cell donor; an ECOG PS ≤2; and adequate hepatic, renal, and cardiac function. The trial uses a 3+3 dose-escalation design, with an initial starting dose of 0.02 mg/kg. Overall objectives are to investigate the safety, tolerability, pharmacokinetics (PK), pharmacodynamics (PD), antileukemia activity, and to establish the minimum safe and biologically effective dose of MGTA-117. The observation period for dose-limiting toxicities (DLTs) is 21 days.
Results: Demographics and disease characteristics of enrolled pts are shown in the Table. In Cohort 1 (0.02 mg/kg), 4 pts were dosed; 1 pt withdrew from the study prior to completion of the DLT observation period because of disease progression and was replaced. To date, 2 pts have been dosed in Cohort 2 (0.04 mg/kg). MGTA-117 has been well tolerated to date; no DLTs, treatment-related serious adverse events (AEs), treatment-related deaths, or treatment-related hypersensitivity or anaphylactoid infusion reactions have been observed. One pt had transient related grade 1 elevations in alanine and aspartate aminotransferase levels that resolved without intervention. The remaining treatment-emergent AEs (TEAEs) were consistent with underlying disease and were considered unrelated to MGTA-117. TEAEs occurring at a frequency of >25% irrespective of causality included nausea, vomiting, tachycardia, thrombocytopenia, anemia, and liver enzyme elevation.
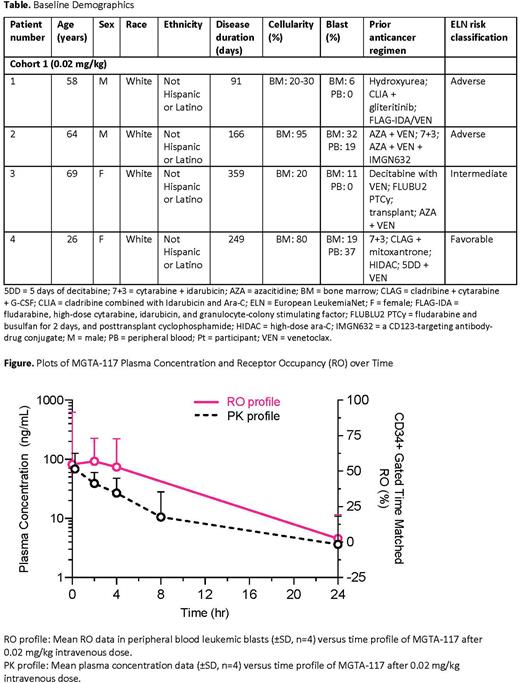
Maximum concentrations of MGTA-117 in Cohort 1 were reached within 1 hour post-infusion. As expected, the observed half-life was approximately 10 hours or less, and MGTA-117 concentrations were no longer measurable 48 hours after dosing in all pts (n=4). Free amanitin-containing payload was undetectable in all PK blood samples from all pts indicating stability of the ADC linker. MGTA-117 was shown to rapidly and selectively bind CD117+ cells in the blood as measured by a receptor occupancy (RO) assay, with reduction of binding to low levels within 24 hours post-dose. MGTA-117 concentrations in blood consistently mirrored RO levels over time (Figure). There was evidence supporting biologic activity of MGTA-117 in the bone marrow in 2 of 3 evaluable pts at the lowest dose level; an observed reduction in CD117+ erythroid progenitors in the bone marrow of 1 pt and complete remission with hematologic recovery (CR) in a second pt with primary refractory AML failing 2 lines of induction chemotherapy. The pt with CR later proceeded to HSCT with successful engraftment. Cohort 2 enrollment is ongoing, and further data are forthcoming.
Conclusions: MGTA-117 has been well tolerated with no unexpected safety concerns. Preliminary data show in vivo stability of the ADC with rapid clearance from blood, robust binding of MGTA-117 on CD117+ cells, and early evidence of single-agent biological activity. The observed PK/PD profile in humans is highly consistent with predictions based on data from studies in preclinical species. The trial continues to advance, and progress is being made toward development of MGTA-117 as a myelodepletive conditioning agent for HSCT in AML/MDS and gene therapy indications.
Disclosures
Westervelt:Pfizer: Consultancy. Kebriaei:Ziopharm: Research Funding; Pfizer: Consultancy; Kite: Consultancy; Amgen: Research Funding; Jazz: Consultancy. Artz:Abbvie: Honoraria; Magenta: Honoraria. McCarthy:Magenta Therapeutics: Consultancy, Honoraria; Bluebird Bio: Consultancy, Honoraria; Juno: Consultancy, Honoraria; Axios: Consultancy, Honoraria; Takeda Pharmaceuticals America, Inc.: Consultancy, Honoraria; Partner Therapeutics, Inc.: Consultancy, Honoraria; Karyopharm Therapeutics Inc.: Consultancy, Honoraria; Janssen Global Services, LLC: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Oncopeptides: Consultancy, Honoraria; Sanofi: Consultancy; Genentech: Consultancy, Honoraria; Fate Therapeutics: Consultancy, Honoraria; Starton Therapeutics: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Bristol Myers Squibb Company: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Stein:PTC Therapeutics and Syros: Membership on an entity's Board of Directors or advisory committees; Auron Therapeutics: Current equity holder in private company; Bayer: Research Funding; Daiichi-Sankyo, Celgene Pharmaceuticals, and Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; PinotBio, Bristol Myers Squibb, Jazz Pharmaceuticals, Foghorn Therapeutics, Blueprint Medicines, Gilead Sciences, Janssen Pharmaceuticals: Consultancy; Astellas Pharmaceutical, Agios Pharmaceuticals, and Genentech: Consultancy, Membership on an entity's Board of Directors or advisory committees; Amgen, AbbVie, Seattle Genetics, and Biotheryx: Consultancy; Syndax: Consultancy, Research Funding. Humphrey:Cyteir Therapeutics: Membership on an entity's Board of Directors or advisory committees; Magenta Therapeutics: Current Employment. Baeder:Magenta Therapeutics: Current Employment. Lee:Magenta Therapeutics: Current Employment. Occhiuti:Magenta Therapeutics: Current Employment. Tang:Magenta Therapeutics: Current Employment. Santos:Magenta Therapeutics: Current Employment. Bertelsen:Magenta Therapeutics: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company; Janssen Pharmaceuticals: Current equity holder in private company; Abbvie Pharmaceuticals: Current equity holder in private company. Mahender:Magenta Therapeutics: Current Employment. Henry:Magenta Therapeutics: Current Employment. Rose:Magenta Therapeutics: Current Employment.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal